Questions: At 70 degrees C, the compound is in the gas phase, and at 20 degrees C the compound is in the solid phase. At 6 minutes, the compound is undergoing the phase change called From 13 minutes to 16 minutes, the compound is in the liquid phase.
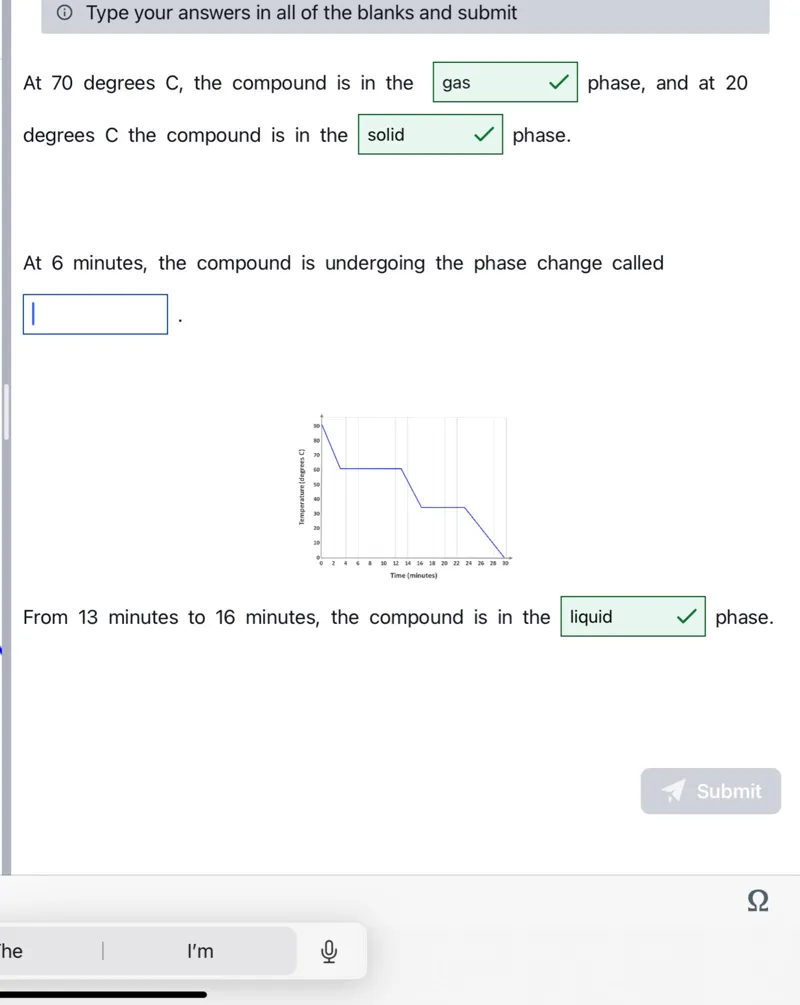
Transcript text: At 70 degrees $C$, the compound is in the gas phase, and at 20 degrees $C$ the compound is in the $\square$ solid phase.
At 6 minutes, the compound is undergoing the phase change called $\square$
From 13 minutes to 16 minutes, the compound is in the $\square$ liquid phase.





